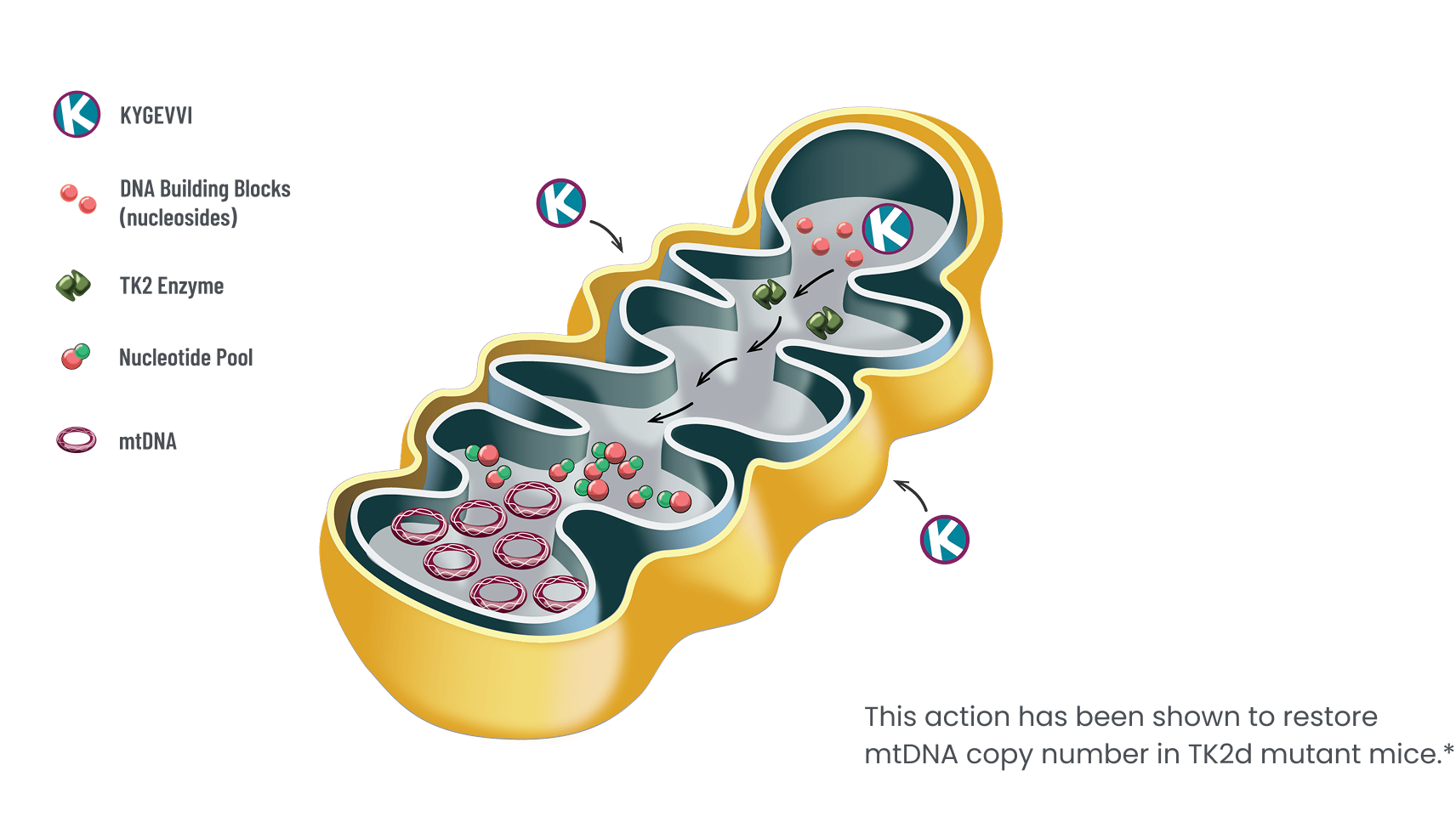
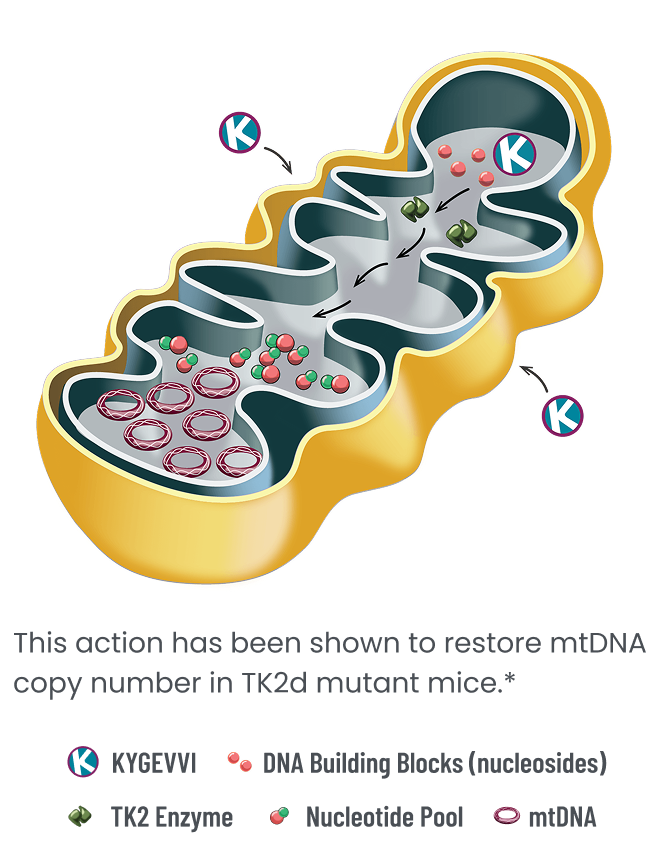
KYGEVVI is indicated for the treatment of thymidine kinase 2 deficiency (TK2d) in adults and pediatric patients with an age of symptom onset on or before 12 years.
Increase in Liver Transaminases
Elevated liver transaminase [alanine aminotransferase (ALT) and/or aspartate aminotransferase (AST)] levels were reported in patients treated with KYGEVVI. Obtain baseline liver transaminase (ALT, AST) and total bilirubin levels in patients prior to treatment initiation with KYGEVVI. If signs or symptoms consistent with liver injury are observed, interrupt treatment with KYGEVVI until liver transaminase (ALT, AST) and total bilirubin levels have either returned to baseline or stabilized at a new baseline value. Consider permanently discontinuing KYGEVVI if signs or symptoms consistent with liver injury persist or worsen. Monitor liver transaminases and total bilirubin levels yearly and as clinically indicated.
Gastrointestinal Adverse Reactions
Diarrhea and vomiting leading to hospitalization, dose reduction, and permanent discontinuation were reported in patients treated with KYGEVVI. Based on the severity of the diarrhea and/or vomiting, reduce the dosage of KYGEVVI or interrupt treatment until diarrhea and/or vomiting improves or returns to baseline. Consider restarting KYGEVVI at the last tolerated dose, and increase the dose as tolerated. For persistent or recurring diarrhea and/or vomiting, consider discontinuing KYGEVVI permanently and provide supportive care with electrolyte repletion as clinically indicated.
Adverse Reactions
The most common adverse reactions (incidence ≥5%) are diarrhea, abdominal pain (including abdominal pain upper), vomiting, alanine aminotransferase increased (ALT), and aspartate aminotransferase increased (AST).
Please see the full Prescribing Information.